Overview of Characteristics
The characteristics of Vitamin C are physically and chemically defined. Despite being a common vitamin and a necessary dietary component, Vitamin C is complex and characterized by many chemical and physical traits.
Chemical Characteristics
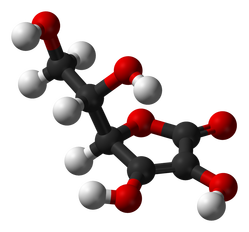 Molecular structure of ascorbic acid (Vitamin C)
Molecular structure of ascorbic acid (Vitamin C)
Also known as ascorbic acid in certain forms, Vitamin C is an organic water-soluble molecule. It is not naturally produced in the human body and is comprised of a 6 carbon molecule base. Five of these carbons form a pentagonal base. Eight hydrogen and 6 oxygen atoms extend from the carbon base. The molecule is polar with an unequal distribution of charges.
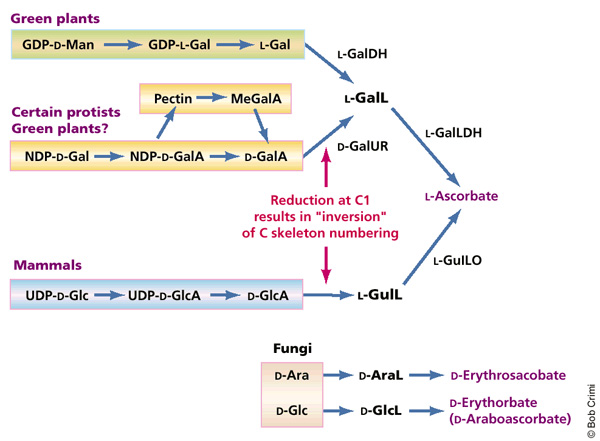
Vitamin C Biosynthesis
Vitamin C is chemically produced through stages of biosynthesis in plants, protists, non-human mammals, and fungi. Mammals capable of synthesis produce Vitamin C from a glucose, but humans lack the enzyme required for Vitamin C synthesis, "l-gulonolactone oxidase." Biosynthesis is a series of catalyzed reactions powered by enzymes to synthesize a product. Biosynthesis involves a complex series of reactions that each produce a substrate for the next stage of biosynthesis. Eventually, the produced substrates result in the formation of Vitamin C for organisms that are capable of synthesis.
Physical Characteristics
The human body does not naturally provide Vitamin C, and it is classified as "water-soluble". Vitamin C weighs 176.1 grams and has a molar mass of 68 grams per mole. Its density measures around 1.694 grams per cubic centimeter with a melting point of 190 degrees Celsius. Vitamin C boils at 553 degrees Celsius.
These manufactured pill forms are most common, but Vitamin C is also synthetically produced in the form of powdered crystalline or liquid forms.
Functions
Vitamin C is naturally located in typical dietary fruits and vegetables for human consumption. The human body is unable to synthesize Vitamin C so it must be obtained from external nutritious sources.

Dietary substances that contain Vitamin C include lemons, oranges, limes, grapefruit, other citrus fruits, kiwi fruit, pineapple, broccoli, brussel sprouts, cabbage, tomatoes, and even potatoes. Of course, the presence of Vitamin C is not limited to these foods. There are innumerable other substances that contain Vitamin C.
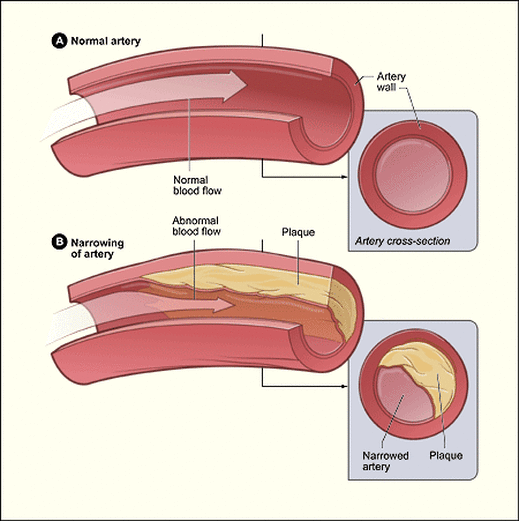
Vitamin C's antioxidant properties are absorbed into the blood steam and clear the arteries of plaque build-up. This prevents cardiovascular issues and heart attacks from occurring.
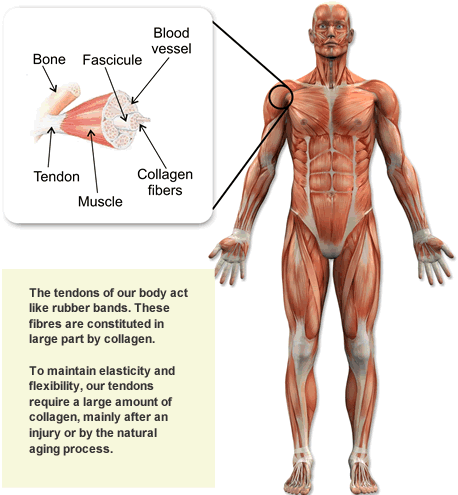
Vitamin C contributes to the formation of collagen by binding cells together and forming a defensive layer of tissue. Tissue plays a significant role in the support of organs and undergoes repair when damaged. Collagen supports tissue to connect tendons, ligaments, skin, bones, teeth, cartilage, blood vessels, and many other body parts. This indicates the significant benefits of the nutrient's bodily functions.
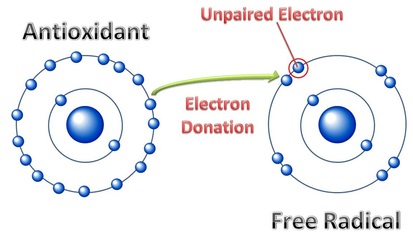
Vitamin C is also a protecting antioxidant against free radicals, molecules that are detrimental to the condition of the body.
Free radicals are unstable molecules because of their unshared electrons. As depicted in the diagram above, antioxidants like Vitamin C offer an electron to make a stable pair of shared electrons. By protecting and preventing bodily damage from free radicals, the body is less likely to age rapidly or experience cancer-causing agents.