Description
Vitamin C can be depicted in various structural diagrams that visualize the molecule's composition. Structural diagrams dissect the molecule to observe Vitamin C in its fundamental form.
Lewis Structure
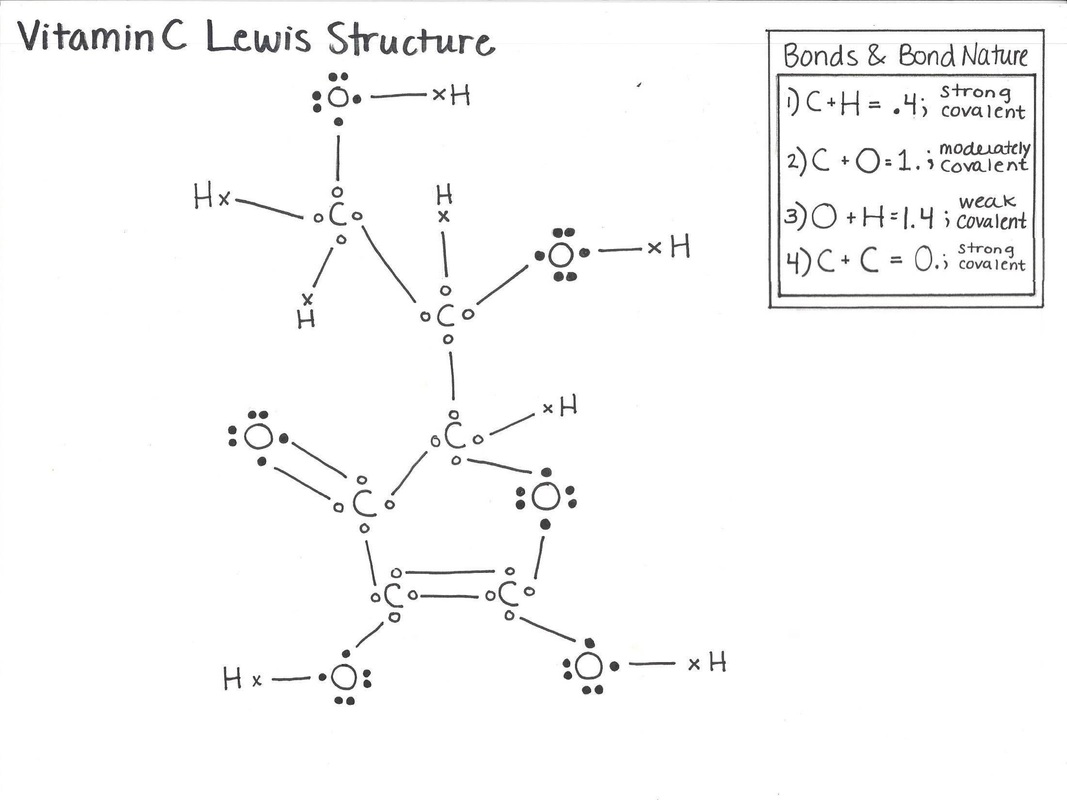
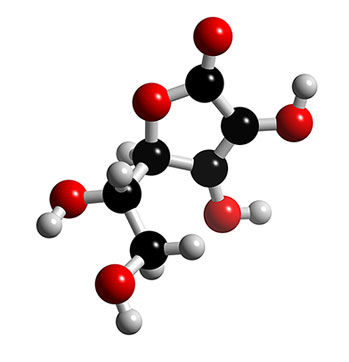
The Lewis Structure for Vitamin C displays the molecule in its skeletal state. The Lewis structure includes the basic elements that comprise Vitamin C and notes the chemical bonds that support the molecule's formation.
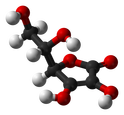
Because the chemical formula of Vitamin C is C6H8O6, the molecular structure is assembled as depicted in the diagrams below. Vitamin C is composed of 6 carbon atoms that function as a basis for the molecule. The appropriate term to define this structural basis is "carbon based", indicating that Vitamin C is a carbon based molecule. Bonded to the carbon base are 6 oxygen atoms and 8 hydrogen atoms.
Lewis Structure diagram
Bond & Bond Natures
Different atoms are bonded together to chemically compose the structure of Vitamin C. The bond shared between carbon and hydrogen atoms differs from the bond between carbon and oxygen. Oxygen and hydrogen share a bond in Vitamin C that varies from carbon bonded to carbon. These bonds possess different natures that can be defined by the term "covalent" or "ionic." By calculating the difference between the electronegativity values of two atoms, one can determine whether a bond is covalent or ionic.
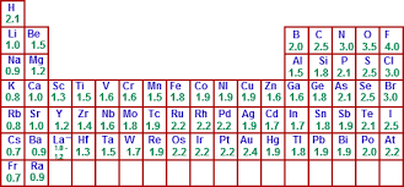
electronegativity chart
Carbon and Hydrogen
According to the electronegativity chart above, carbon has an electronegativity value of 2.5 and hydrogen has an electronegativity value of 2.1. These two values must be subtracted to determine the electronegative value of the bond. By subtracting the values it becomes evident that the bond value is .4. Any bond above the electronegative value of 1.70 is considered "ionic" concluding that the bond between carbon and hydrogen is strongly covalent.
Carbon and Oxygen
The electronegative value for carbon is 2.5 and 3.5 for oxygen. After calculating the difference, it is evident that the bond between carbon and oxygen has an electronegative value of 1.0. This classifies as moderately covalent because it is less than 1.70.
Oxygen and Hydrogen
Oxygen has an electronegative value of 3.5 and 2.1 for hydrogen. The difference between the electronegative values calculates to 1.4 meaning that the bond shared is weakly covalent. As the electronegative value of the bond approaches 1.70, it becomes weaker in covalence.
Carbon and Carbon
Because carbon and carbon are the same element and share the same electronegative value, the difference is 0 indicating that the shared bond is extremely covalent.
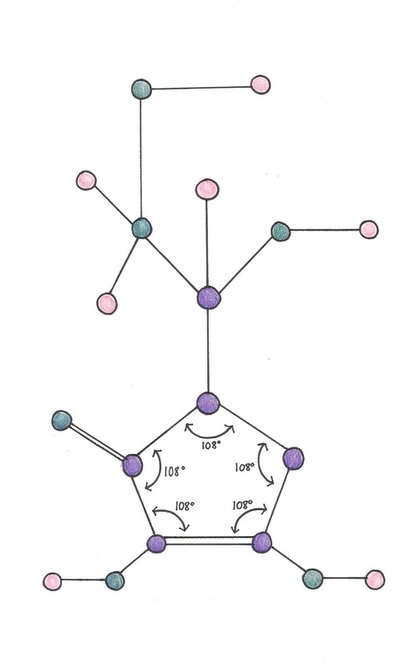
Molecule Structure & AX Form
'AX form' is the geometric form of a molecule that can be determined by referring to a VSEPR chart.
Vitamin C does not have an identifiable AX form because it is cyclically shaped. A pentagonal base of carbon supports the molecule with interior angle measurements of 108 degrees. The other angle measurements of Vitamin C are nonspecific and do not have defined measures.
Vitamin C does not have an identifiable AX form because it is cyclically shaped. A pentagonal base of carbon supports the molecule with interior angle measurements of 108 degrees. The other angle measurements of Vitamin C are nonspecific and do not have defined measures.
Molecular Structure Diagram
Intermolecular forces
Forces that attract or repulse adjacent molecules are termed 'intermolecular forces'. Vitamin C must be located in the general vicinity of another adjacent molecule for intermolecular forces to be observable.
The main types of intermolecular forces include dispersion, hydrogen bonding, and dipole-dipole. The intermolecular force of dispersion occurs between any two adjacent molecules. Hydrogen bonding is an intermolecular force present between the hydrogen atom of one molecule and the oxygen, nitrogen, or fluorine atom of another. Dipole-dipole is the intermolecular force that acts between any two adjacent polar molecules.
The main types of intermolecular forces include dispersion, hydrogen bonding, and dipole-dipole. The intermolecular force of dispersion occurs between any two adjacent molecules. Hydrogen bonding is an intermolecular force present between the hydrogen atom of one molecule and the oxygen, nitrogen, or fluorine atom of another. Dipole-dipole is the intermolecular force that acts between any two adjacent polar molecules.
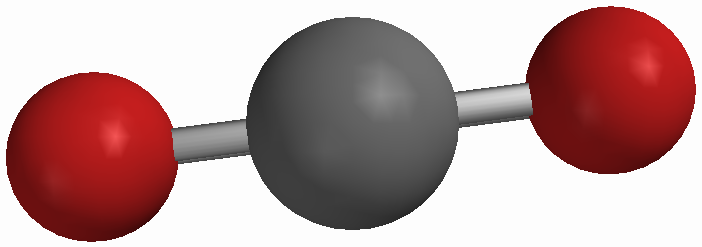
Vitamin C & Carbon Dioxide
|
The intermolecular forces acting between Vitamin C and carbon dioxide include hydrogen bonding and dispersion. Hydrogen bonding is present because hydrogen from Vitamin C is forcefully attracted to oxygen of carbon dioxide.
Dispersion is another present intermolecular force because it occurs between any two adjacent molecules. Dipole-dipole is not an active force between the two molecules because carbon dioxide is nonpolar unlike the polar molecule, Vitamin C.
|
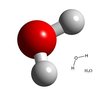
Vitamin C & WATER
|
Dispersion and hydrogen bonding are intermolecular forces that act between Vitamin C and water. Dipole-dipole is not present between the two molecules because water is nonpolar and Vitamin C is polar. Dispersion occurs between any two adjacent molecules, including Vitamin C and water. Hydrogen from Vitamin C is attracted to oxygen from water or vice versa.
|